An observational study for left atrial appendage closure supports CLOSURE AF data
Spring weather and travel attacked Sensible Medicine this week. Sorry for the missed post on Monday. I was at a meeting in France and forgot what day it was.
Today we have rising star, Dr. Mohammed Ruzieh summarizing his recent observational study on left atrial appendage closure in a typical older Medicare population. He’s honest about the study’s limitations, but the findings do two things: a) make you think, and b) reduce enthusiasm for the safety of this procedure. JMM
On the matter of knowledge: In medicine, the most rigorous way to assess the efficacy or harm of an intervention is a randomized trial. In this design, patients are randomly assigned to receive either the investigational treatment or the standard of care, ensuring equal probability of allocation. This randomization minimizes bias and balances both known and unknown confounders between groups, making it the gold standard for determining causal effects.
What are confounders? Confounders are variables that are associated with both the exposure (treatment) and the outcome, and can distort the true relationship between them if not properly controlled. For example, people who carry lighters have a higher rate of lung cancer than people who do not. So, does carrying a lighter cause lung cancer? No, smoking is the confounder. It causes both the behavior of carrying a lighter and the development of lung cancer. The association between lighters and lung cancer is real but spurious, entirely explained by the confounding variable of smoking.
Randomized trials are time-consuming, costly and often enroll healthier patients. Because of these limitations, observational studies are sometimes conducted, as they allow evaluation of large populations in real-world settings. However, their major limitation, especially when comparing treatment versus no treatment, is residual confounding. Treatment decisions are influenced by multiple patient- and physician-level factors; while some can be measured and adjusted for, others are unmeasured or unknown, leading to potential bias. Despite this, observational studies remain valuable for hypothesis generation and real-world evidence.
Today, we discuss our recently published study comparing Watchman versus oral anticoagulation.
Background
What is Watchman? A percutaneous left atrial appendage occlusion (pLAAO) device designed to occlude the left atrial appendage. (Pictures like this belie the complexity of the human body).
What is oral anticoagulation? Medications taken by mouth (e.g., warfarin or direct oral anticoagulants like apixaban or rivaroxaban) that reduce blood clot formation.
Patients with atrial fibrillation can have a fivefold increased risk of stroke, and physicians often prescribe anticoagulation to these patients. Anticoagulants also increase the risk of bleeding. But in patients with enough stroke risk, net benefit occurs because stroke reduction is larger than bleeding increase.
In some cases, the stroke originates from a structure in the heart called the left atrial appendage, a small, pouch-like extension of the left atrium where blood can pool and form clots. The Watchman device occludes this appendage, potentially lowering stroke risk and reducing the need for long-term oral anticoagulants in an effort to reduce bleeding.
The Study
Our study compared pLAAO with oral anticoagulants in Medicare patients with atrial fibrillation to evaluate which approach may better prevent stroke. We included 3,692 patients in the pLAAO group and 11,076 in the oral anticoagulants group (14,768 total – compare this to the largest trial of pLAAO, CHAMPION-AF which included 3,000 patients).
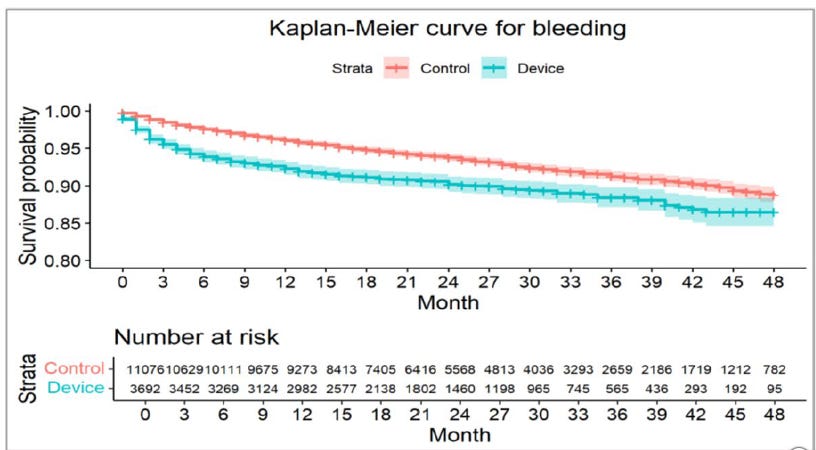
We found that patients who received pLAAO experienced more bleeding leading to hospitalization (5.30/100 person-year vs. 3.25/100 person-year, HR: 1.58, 95% CI: 1.38 – 1.79) but similar rates of stroke and death.
Discussion
We acknowledge that residual confounding may still be present and that our study has limitations, as outlined in our manuscript. Nonetheless, several key findings stand out and support causality.
First, mortality rates were comparable between the two treatment groups, reflecting results seen in randomized trials and reinforcing that we adequately addressed many of the main confounding factors.
More importantly, the mortality rate in our study was approximately 8.4% per year compared to 4.6% over 3 years in the CHAMPION-AF trial. Why is this important? Because CHAMPION-AF didn’t enroll the typical higher-risk Medicare Watchman-recipient and its findings may not apply to them.
Older high-risk patients have more reasons (competing causes) to get stroke other than the left atrial appendage (such as atherosclerosis) and a localized intervention is less likely to be helpful. Furthermore, these patients may be more likely to experience complications from device implantation.
Second, the higher bleeding rate observed with pLAAO in our study is noteworthy. This can be explained by the fact that patients typically receive intensified “blood-thinning” therapy in the first few weeks after device implantation to prevent clot formation on the device, which increases bleeding risk
Additionally, patients typically continue aspirin indefinitely after pLAAO, which may not be safer than apixaban. The device implantation itself can cause bleeding too. While early separation of curves favoring the intervention in observational studies could reflect residual confounding, a point we previously discussed, this is unlikely in this case.
Why? In the case of pLAAO, the early signal of increased bleeding is explained by the procedural risks of device implantation and the subsequent intense anticoagulation regimen. See the figure below from CLOSURE-AF trial, which demonstrates early separation of the curves too. Procedural interventions inherently carry risks, and it is not uncommon to observe initial harm following such procedures.
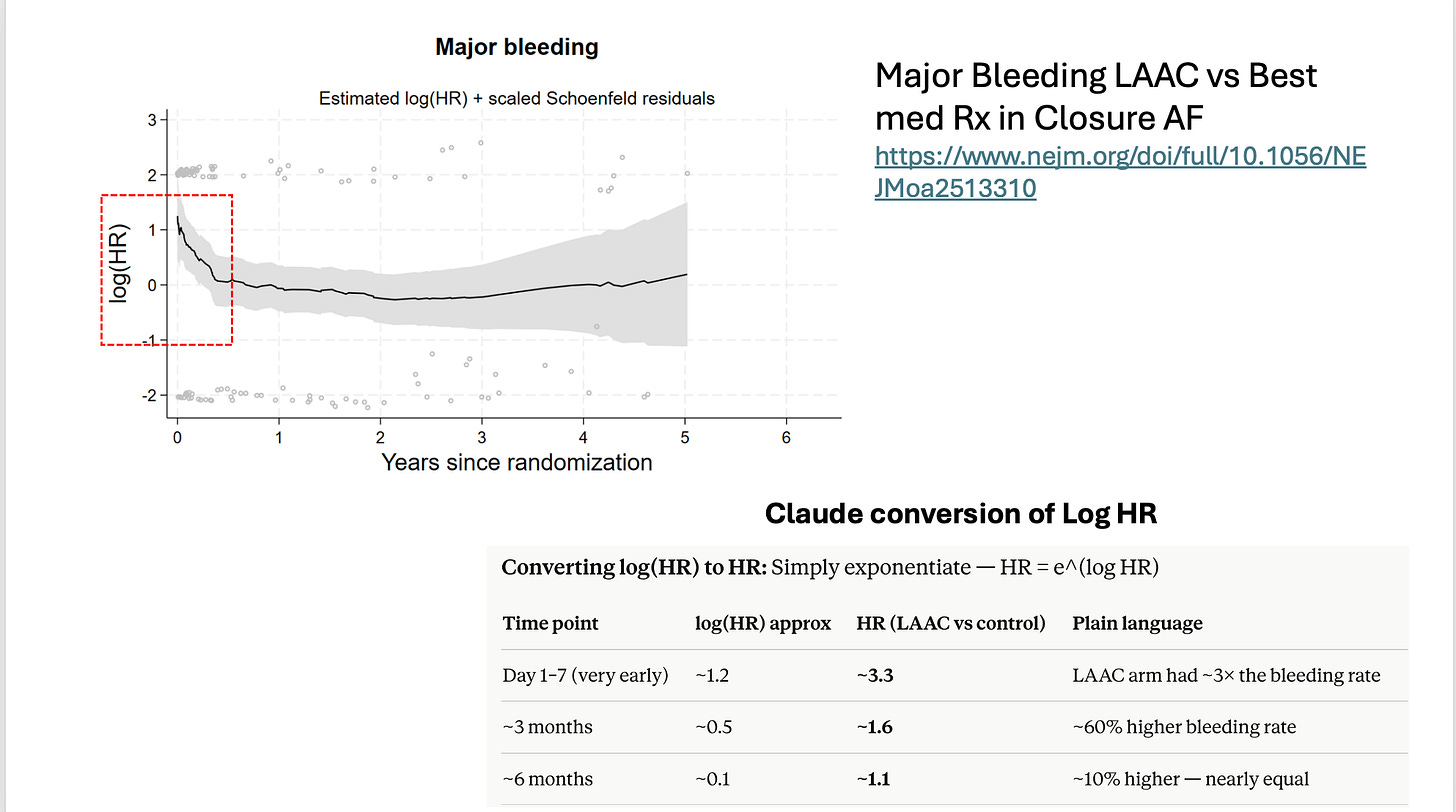
Finally, the results of our study show a similar direction of signal as the randomized CLOSURE-AF trial, which also included a high-risk population. Since our study was conducted prior to CLOSURE-AF, this consistency further strengthens our confidence in the findings.
In summary, while observational studies have inherent limitations, they can still provide valuable insights. In this case, the real world data support the notion that pLAAC does not reduce bleeding largely due to the early increase in bleeds. It also supports the RCT observations that older more ill patients likely garner less net benefit.
Dr Ruzieh is too nice to say this but I will. This observational study was rejected by numerous journals before landing in the Journal of Interventional Cardiac Electrophysiology. You might argue that it was rejected because it was an observational nonrandomzied comparison study, which are often marred by bias. That’s possible, but in the many months it was being rejected at various journals, these same journals published a slew of other similar observational studies. Publication bias is not obvious but is important to consider when reading the medical literature. JMM




Thank you, Dr. Ruzieh. Excellent.
Disclaimer: I am not a Dr. and I did not stay at a Holiday Inn last night. However, I read quite a few Sensible Medicine articles, as well as other medical Substacks and sometimes even underlying studies. I don't have AF, but am fascinated by the condition, including why the LAA even exists ...
First, Dr Ruzieh: Wonderful article and your ability to write in terms that are easy to comprehend by mere mortals is greatly appreciated. Write more!
Second, Dr. Mandrola: Your note at the end is also really appreciated. It is a depressing reality that so much of what constitutes "the literature" is corrupted by publishing house bias. From a potential patient perspective for any medical intervention, it is just a tad unnerving.