Is the FDA too lax: Part 2
I started it all by claiming on Twitter that the FDA was a rubber stamp. From boosters for 6 month old babies (no data), to postpartum depression drugs that are basically Xanax, to bad cancer drug approvals, in my mind, the FDA is failing the American people.
In a recent post, John Mandrola reviewed 5 cardiology devices approved by the FDA with questionable data. Here, I want to give some oncology examples to show how low the bar is.
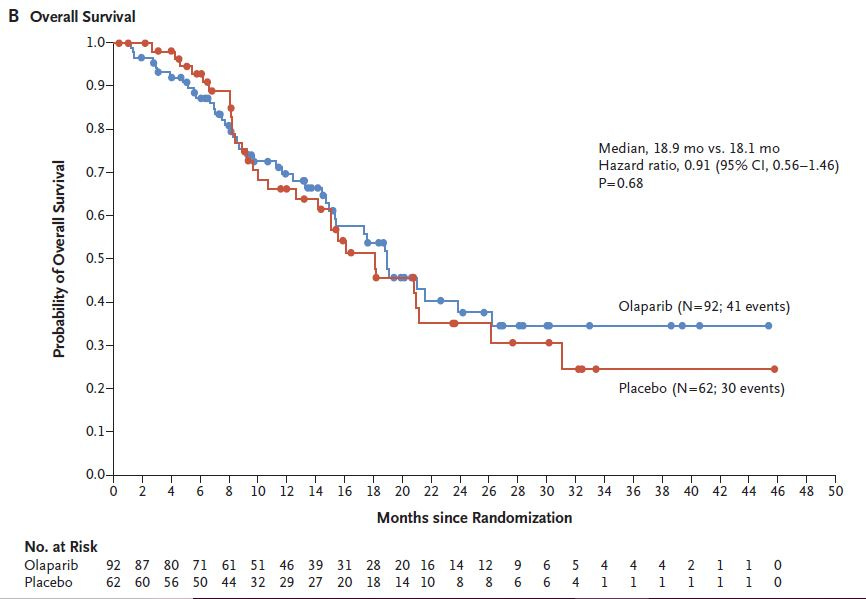
Olaparib in pancreas cancer
The US FDA granted approval to this $12,000 a month cancer pill based on clinical trials where it improved progression free survival over placebo. Here is what the drug did for survival.
And there was no quality of life benefit. Finally, you may be asking. Wait, is placebo really the appropriate control arm for pancreas cancer? That doesn’t sound right. That’s because it isn’t — placebo control was unethical.
I discuss this trial at minute 39 here.
Fruquintinib
In 2023, the FDA approved this drug in a placebo controlled trial in colon cancer.
The drug improved survival, which is better than Olaparib; but once again placebo control was unethical.
Here is proof. First, patients had not seen Regorafenib or Trifluridine-tipiracil, two existing treatments. These should have been part of an investigator choice control arm.
Second, after the trial ended, patients received other drugs that also should have been available.
Had Fruquintinib been tested against the actual standard of care, I doubt it would have won.
ADAURA lung cancer
The FDA now allows patients whose cancer has been cut out to take Osimertinib for years. This $22,000 a month medicine supposedly improved survival in the adjuvant setting. But, the study does not use US staging (which enriches for occult advanced disease and makes it more likely to win). The study does not provide Osimertinib upon recurrence — which was already the US standard of care — and also distorts the trial. And, many people skipped adjuvant chemotherapy — another standard of care that is already proven. I outline all the errors here, but the bottom line is I have no idea if giving Osimertinib after surgery is better than reserving it for the people who relapse (a less toxic and cheaper option).
Cancer drugs are technical
The truth is cancer drug approvals are often technical, and have hidden pitfalls. I try to explain these on my podcast Plenary Session. But the net result is most drug approvals in oncology have no relevance for the average American with cancer. We spend more money, and companies get rich, but patients and the public gets hosed. I think the FDA needs a real reform if it is to serve its purpose, and that is why I tweeted.








It is time to defund the FDA, CDC, NIH and others.
Conflicts of interest have run amok.
I don't work for the FDA, but I do work for another government regulatory agency. I don't feel up to the task of describing the level of dysfunction taking hold in Federal civil service, but I'll attempt to summarize one aspect of it. Things are growing progressively worse as staff loses the distinction between being a political appointee who advances the message of the current administration-any administration-and being civil service, charged with impartially executing the law. What matters now is pleasing the right people, always saying yes, and doing what you're told-even if it's wrong. I imagine that the regulations governing FDA approvals are actually fairly decent, but the idea of someone internal insisting that an agency just follow the regulations is becoming radical. The government is slipping into regulation by feelings. And it probably feels pretty good to the agency higher-ups to approve all these money making drugs and devices.