Study-of-the-Week: A Critical Appraisal of Renal Denervation for High Blood Pressure
Academic cardiologist Andrew Foy explains why we should remain skeptical regarding renal denervation for the treatment of high blood pressure
Dr. Andrew Foy returns to Sensible Medicine with a critique of renal denervation, a novel approach to treating high blood pressure (hypertension).
Renal denervation is a procedure in which a doctor places a catheter into the femoral (leg) artery up to the kidney and then ablates (burns) the nerves going to and from the kidney. The idea is to reduce the input of the adrenergic (think adrenaline) nervous system (aka the sympathetic nervous system) on the kidney. Many patients with high blood pressure have abnormally high sympathetic tone.
The potential of this procedure is massive. Hundreds of millions of humans have high blood pressure, which in turn increases the odds for serious complications, such as stroke, heart failure, and kidney failure. What’s more, adherence to blood-pressure-lowering interventions (lifestyle measures and tablets) is imperfect.
A one-time procedure that could reduce blood pressure over the long-term would represent a major medical advance, and bring riches to all those involved.
The early renal denervation data looked amazing. Then a proper trial crushed the early enthusiasm. Proponents did not give up; newer studies using improved techniques have shown some promise. Now, though, there is a push to use weaker data to over-promote renal denervation. If the proponents succeed, it’s likely that doctors would embrace this easy-to-do procedure.
Foy argues, persuasively, that the newer data is not strong enough to unleash this procedure into the wilds of American healthcare. The best part of his column is that it transcends one specific medical topic and educates us on how to spot biases in medical trials. John Mandrola
Don’t Be Fooled by the Latest Renal Denervation Claims
by Andrew Foy, MD,
I’m starting this post with a picture because you know how the saying goes (Figure 1).

Let’s start with a bit of history. SYMPLICITY HTN 2 was the first randomized trial to compare patients undergoing renal denervation (RD) to a control group of patients who did not undergo the procedure.
All participants had treatment resistant hypertension. The patients, personnel and outcome assessors knew the treatment assignments. The pre-specified primary endpoint of the trial was the difference in office systolic blood pressure (SBP) at 6 months.
The results were stunning: on average, patients assigned to RD experienced a 32-point drop in their SBP compared to a 1-point increase for those assigned to the control group.
The authors’ interpretation reads:
“Our randomized, controlled trial confirms the role of renal sympathetic nerves in essential hypertension and validates a new therapy for treatment-resistant hypertension.”
The problem with SYMPLICITY HTN 2 was its unblinded nature. Participants and personnel were not blinded, nor were the outcome assessors.
When participants and personnel are not blinded in a randomized trial, it is at high risk for performance bias. Performance bias may occur if one group of subjects in an experiment is treated differently by investigators due to knowledge of their assignment. It may also occur if one group of subjects acts differently in an experiment due to knowledge of their assignment.
The key takeaway is that these departures, whether on the part of the investigators or patients, may affect the outcomes of the study but are not due to the intervention being studied.
But that’s not all. When those who assess outcomes are not blinded, studies are at high risk for detection bias. Detection bias refers to systematic differences between groups in how outcomes are determined. Again, it is the difference in how outcomes are determined that affects the results of the study and not the intervention being studied.
While it is impossible to say whether these biases affected the results of SYMPLICITY HTN 2, or, for that matter, any study that does not employ blinding, it is reasonable to be skeptical, and ask for higher quality studies to reduce or eliminate the possibility that biases in conduct affected the results.
This is why the SYMPLICITY HTN 3 trial was performed.
In SYMPLICITY HTN 3, patients with severe resistant hypertension were randomly assigned to undergo RD or a sham procedure. Before randomization, patients had to be receiving at least 3 BP-lowering drugs at maximally tolerated doses, including a diuretic. The primary endpoint was the difference in office SBP at 6 months.
Patients were unaware whether they underwent RD or the sham procedure and the authors made the point of specifying in the methods that “blood pressure assessors were also unaware of the study group assignments.”
What a difference this made: at 6 months, there was no significant difference in the change in office SBP. The difference between the RD and sham controlled group was -2 mmHg (-14 vs -12 mmHg); it was -33 mmHg (-32 vs +1 mmHg) in the unblinded SYMPLICITY HTN 2 trial.
The authors sobering conclusion was that more rigorously designed trials would be needed to validate alternative methods of RD or to confirm previously reported benefits in patients with resistant hypertension.
Following SYMPLICITY HTN 3, at least 9 additional RCTs have tested RD against a sham control. These trials are covered in a meta-analysis by Ahmed et al, which I was the senior author on, and whose results are included in the opening figure.
We found that compared to sham control, RD significantly reduced office BP (-4/2 mmHg), 24-hour ambulatory (-3/2 mmHg) and daytime ambulatory BP (-4/2 mmHg) but not nighttime BP.
We concluded that compared to sham control, RD significantly reduced most measures of BP; however, reductions are modest and within the bounds of what can be expected by standard BP-lowering medications.
At this point, you may think that the conclusions regarding the efficacy of RD as a blood-pressure lowering intervention are pretty clear: “the highest quality studies show a small but significant reduction over the short-term.”
What you would probably not think is, “we should conduct more testing of the BP-lowering efficacy of RD by using lower quality study designs.”
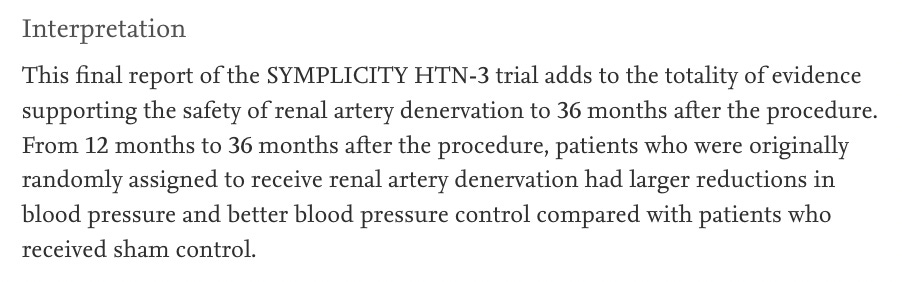
But this is exactly what the latest study from the SYMPLICITY HTN 3 investigators does. Bhatt et al. report, in the influential journal, The Lancet, that at 36 months, the final change in office SBP was -22 mmHg in favor of the RD group.
Recall that in the original (sham-controlled period), there no significant changes over 6 months and now there were large differences at 3 years? That is curious.
I could now engage in mental acrobatics to explain this curiosity.
The simple reason is that after 6 months, which was the pre-specified primary endpoint of the original trial, patients were unmasked. (It’s unclear whether BP assessors were as well; I’d assume so.)
See the red box in the figure below from the follow-up study.
Of the 364 patients originally assigned to RD and the 171 assigned to sham control only 219 (60%) and 134 (78%) were included in the 36-month analysis. Of the 134 original sham control patients, the overwhelming majority of these had BP values for 36 months imputed from their last measured BP before crossing over to receive RD.
This begs the question, “Why did patients need to crossover given the difference between masked BP between the RD and sham groups was -2 mmHg?” but that would be a digression.
The eminent authors concluded in The Lancet
At the end of the day you have to ask yourself, “How seriously should I take these latest claims?” My answer would be, “forget about em.” There was no blinding.
A hierarchy of research methodology exists for a reason; mainly to reduce the risk of bias. In the case of RD, we have data from high quality designs that provides one answer and data from lower quality designs that provides another (Figure 2).

This is easy folks! Ignore unblinded data.
The only way to answer the question of whether this procedure works over years not months is to do more trials with longer follow up periods while everyone is blinded. They could allow med up-titration and additions to take place if BP exceeds some limit. Will this be done? We shall see.






Excellent Dr. Foy. Thanks so much.
loved this the placebo affect in full force thaks dr. foy