What happens when doctors run unethical trials? They get promoted
The CONTACT-2 study
What happens to doctors who run unethical clinical trials?
Ans: they get praised and promoted
In this essay, I am going to explain to you why the recent study CONTACT-2— presented now at a cancer conference— is a problematic trial. Next, I am going to discuss why these trials persist.
The reason is that the investigators are rewarded not punished.
Part 1: Why is CONTACT-2 problematic?
CONTACT-02 is a global, randomized trial testing two costly, new, toxic drugs Cabozantinib (the drug from hell) and Atezolizumab. In the trial they test it against abiraterone+pred or enzalutamide.
Randomized trials are amazing, but the design matters. The control arm has to the best available care that you would give the patient if they were not in the study. The trial should be designed such that if your father was assigned to the control arm, you would be reassured knowing he was getting the best proven care absent the experimental drug. You can’t randomize him to therapy worse than what he would otherwise get.
This trial failed to do that. The control arm is known to be deficient. Abi and Enza are good drugs in prostate cancer, unless you have already have gotten one or the other.
As I wrote in 2021 for a similar trial, “The control arm of abiraterone or enzalutamide… is suboptimal since there is substantial cross-resistance between these agents, leading to poor response rates and short PFS [3], [4], [5].”
Giving these drugs over again happened in 100% of people here.
In this case, they are known to work poorly. A good doctor would instead give many other proven options: docetaxel, cabazitaxel and perhaps even Lu177 PSMA, but not these drugs again! Yet, the investigators banned those choices. Even the discussant notes this (PS: it is rare in cancer meetings to invite a discussant who is actually critical).
After they progressed on this trial (i.e. after they had their time wasted), a small percentage did get those good drugs, but not as high as if the trial had been run solely in the US. Even the discussant raises the point that giving the good drugs after the trial ended might even be too little too late.
CONTACT-2 is in line with other unethical trials. The same problem happened with the PROFOUND study, detailed here.
Many others agree
Finally, we have documented this occurs often in trials submitted to the US FDA. It occurs in 17% of all trials accepted by FDA!
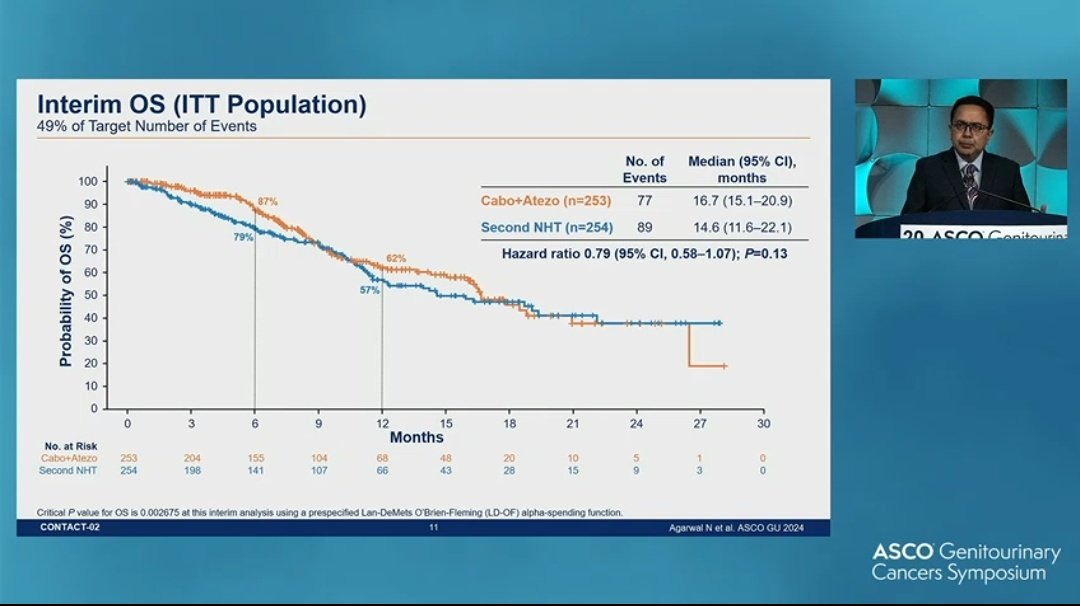
Despite all that the drug barely works! Here was the surrogate endpoint PFS
And here is overall survival. Totally null!
Part 2: What happens to these investigators?
Having established the control arm was beneath global best standard of care, and not giving the good drugs hastened death in the control arm. It’s clear that Cabo+Atezo is not just a costly, toxic drug combo that doesn’t improve survival. If it were approved it probably would WORSEN overall survival b/c it would be given in lieu of better choices. The patients in the control arm were harmed by a problematic trial.
So what happens to the people who signed off on the study?
They are happy about it! They celebrate their oral presentation!
Their colleagues cheer them!
I am stunned that doctors could praise a trial that harmed the control arm participants— so gravely— by denying the best available therapy outside to the study. To be it is an abdication of the duty of a doctor. It is when careers take precedence over ethics.
As for promotion…
What does this all mean?
Oncology is doomed if we reward unethical trials with oral plenary sessions, and lavish, obsequious praise. Junior colleagues are learning— tacitly— to butter up the trialists whose choices directly harm cancer patients. These drugs are already approved and can be used off label. That might even be worse than not having this choice! Even with a flawed control arm, they only marginally improve a useless surrogate endpoint.
I have more thoughts, so subscribe to my Youtube channel for a video on this topic soon.
What do you think? How can we stop this? Leave a comment
















Same thing Brandeis tried to do a 100 years ago: If the broad light of day could be let in upon men’s actions, it would purify them as the sun disinfects.
Thank God for Covid. It took away the veneer of trust I had in CDC/FDA/most Doctors. Who knows, maybe I'd be on Ozempic were it no for Covid opening my eyes (actually, I wouldn't because I'm not *that* stupid) So while I'm not an SME and have no idea what the specifics are, it reads like par for the course. If NIH and CDC and FDA behave unethically, why WOULDN'T everyone else?
I think it would be appropriate to contact the Ethics committees that approved these studies and the Heads of their institutions to ask them to review their process of reviewing studies to ensure that they are upholding the requirement for best available therapy in the control arm.