When Is the Evidence Enough?
The Case of Anal Cancer Screening
We obviously spend a heap of time on Sensible Medicine considering screening tests.1 We do this because there is little in medicine where the disconnect between what should work and what has been shown, in robust studies, to work is as great. We also write about screening tests often because new ones seem to be proposed almost monthly. Some of these — Grail, full body MRI — are now advertised directly to people.
Today, I am pleased to share an article by Ken Noguchi in which he considers the data behind yet another screening intervention.
Adam Cifu
A primary care physician must decide how much evidence is enough before recommending a screening test. If we cannot expect every screening modality in every subpopulation to have multi-site RCTs with long-term follow-up, how do we decide how much evidence is enough? This is where we are with anal cancer screening.
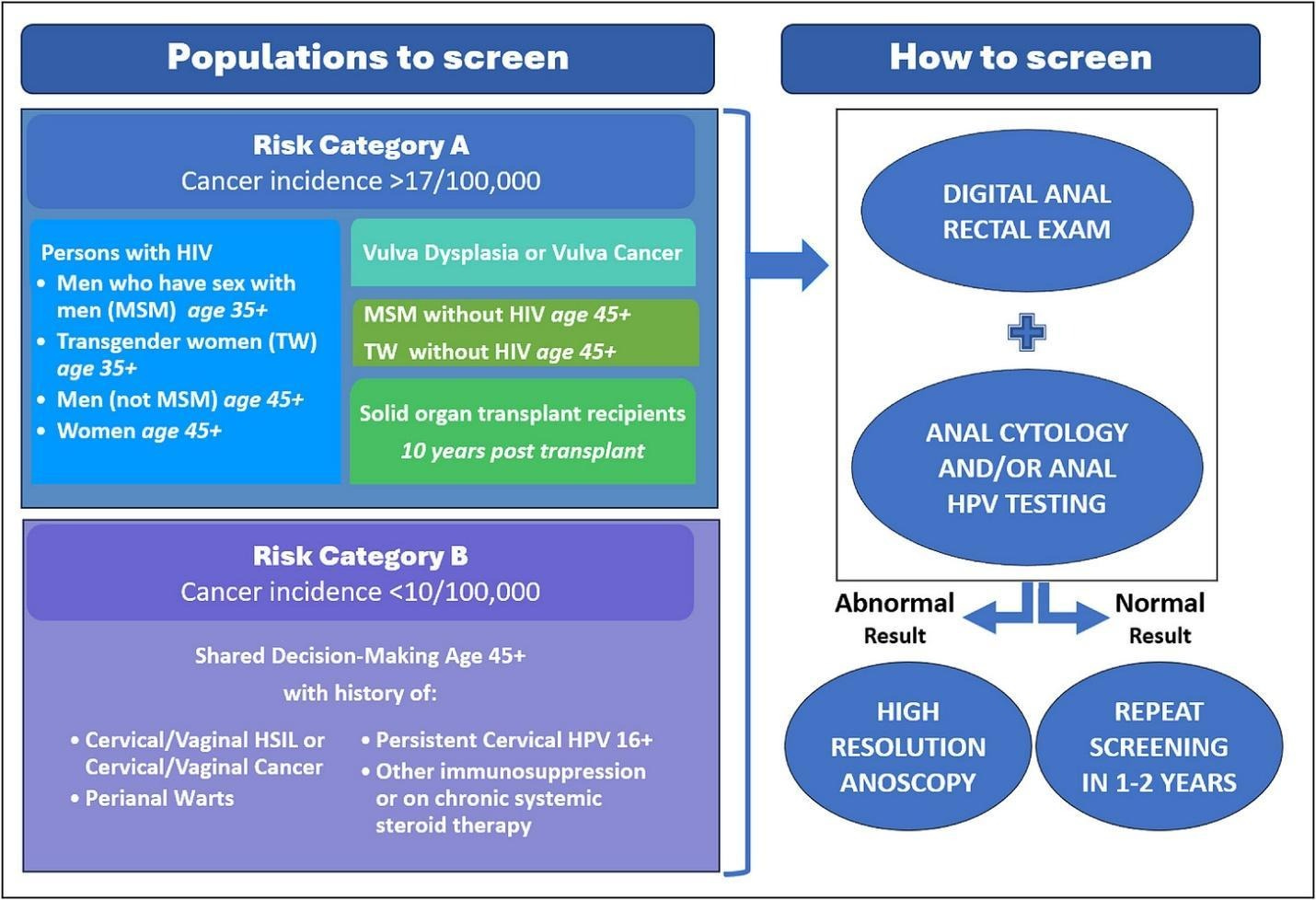
Guidance for anal cancer screening is sparse. It’s only addressed by one guideline, written by the International Anal Neoplasia Society (IANS). Here is the infographic summary of their recommendations:
The recommendations are mostly based on epidemiologic studies that identify risk factors increasing the incidence of anal cancer. Men with HIV who have sex with men have an anal cancer incidence of 85 per 100,000 person-years, men without HIV who have sex with men have an incidence of 19 per 100,000 person-years, and the general population has an incidence of 1-2 per 100,000 person-years. The authors propose screening populations whose anal cancer risk is 10-fold greater than that of the general population. They also cite the ANCHOR trial:
Study Results
The ANCHOR trial enrolled patients with HIV who were at least 35 years of age and had biopsy-proven high-grade squamous intraepithelial lesion (HSIL) of the anus. 4459 patients were randomized to clinician-determined treatment of HSIL (ablation, excision, topical treatments) or active surveillance. They found a statistically significant reduction in anal cancer incidence from 0.9% in active surveillance patients to 0.4% of treatment patients. There were 48 deaths in the active surveillance group and 55 in the treatment group, a non-significant difference. The researcher identified 1 trial-related serious adverse event (including pain and infection) in the active surveillance group and 7 in the treatment group.
Comments
The ANCHOR trial is rigorous and reliable, but we must be careful not to overextrapolate. ANCHOR demonstrates that patients with HIV and HSIL of the anus benefit from treatment of HSIL by preventing cases of anal cancer. It does not demonstrate that anal cancer screening prevents anal cancer incidence or mortality. It also only includes patients with HIV.
My conservative instinct is to push back and say that we cannot recommend this without an RCT of screening. Dr. Paul Sax expressed similar concern regarding anal cancer screening in HIV. But am I right to demand RCTs for every screening test, in every population?
Cancer screening is an inherently probabilistic activity. We estimate a patient’s pretest probability of cancer, update it based on a screening test result, and then recommend further tests. In a population at low risk for cancer, even using a test with excellent sensitivity and specificity, screening leads to huge numbers of false positives, with all the downstream harms and costs. Conversely, in high-risk populations, more of the positive tests will be true positives.
The question is not whether risk matters to cancer screening; it clearly does. The question is whether we should accept a low level of evidence before deciding to screen high-risk populations. There are examples where we’ve adopted cancer screening in high-risk populations without direct RCT-level evidence.
One example is liver cancer screening in patients with cirrhosis. Because liver cancer is rare, we do not recommend universal screening. However, for specific high-risk populations such as patients with cirrhosis, the AASLD guideline recommends screening with AFP and ultrasound despite having no RCT-level evidence. This recommendation is based on epidemiological studies that show an increased incidence of liver cancer with cirrhosis: the incidence of liver cancer with cirrhosis is roughly 2,000 per 100,000 person-years, in the general population, the risk is only 5 per 100,000 person-years. The authors also cite a related RCT of liver cancer screening in patients with hepatitis B, which showed a 37% reduction in liver cancer mortality.
In the case of liver cancer screening in cirrhosis, there are direct RCTs. We have adopted screening based on the RCT of screening patients with hepatitis B and the fact that cirrhosis increases the risk for liver cancer by over 100-fold compared with the general population.
Contrast this with the case of anal cancer. There are no RCTs of anal cancer screening. The closest trial we have shows that treating precursors of anal cancer reduces the incidence of anal cancer. I see the potential, but for now the evidence feels too limited for me to recommend screening, even in high-risk populations. Yet, the comparison to liver cancer screening in cirrhosis raises more questions. At what point does increased risk justify extrapolating from indirect evidence? How indirect can that evidence be? And how do we ultimately decide when the evidence is sufficient?
Ken Noguchi, MD, PhD is an assistant professor with the University of Wisconsin Department of Family Medicine and Community Health. He writes a Substack about evidence-based primary care.



I am so excited about the website collection of screening articles. Thank you!
I guess it's a good thing that Sensible Medicine and the internet didn't exist in Dr. Papanicolau's day - the Pap test clearly wouldn't have made it out of the gate . All those pelvics and smears every year on every woman most of them normal , but a boat load of false positives - and we didn't even know what caused cervical cancer at that time. The nice thing about science is that screening can be refined over time - as it has with cervical cancer. And lots of women alive because of a screening program that in retrospect would have been described as flawed and overreaching .And the whole idea behind the screening was that this was a terrible disease and deserved our attention . But if you never screen, then there is no refinement , just a shoulder shrug " well nothing we can do" . Don't get me wrong , I am not a fan of a lot of " routine " testing - but I think we should avoid adopting a nihilistic philosophy about it as well -