When Surrogate Endpoints Best the Primary Endpoint
Patients with respiratory failure benefit from supplemental oxygen. Oxygen can be delivered through a specialized high-flow nasal cannula or standard cannula. High-flow oxygen is remarkable because everyone in the room can tell it is flowing like crazy.
Eleven years ago, the FLORALI trial found that high-flow oxygen improved mortality vs standard flow oxygen (HR 2.01 95% CI 1.01-3.99) and non-invasive ventilation (HR 2.50 95% CI 1.31-4.78). High-flow oxygen also reduced intubations and improved breathlessness. During the COVID pandemic, high-flow became accepted practice and guideline writers endorsed its use.
Yet the FLORALI trial randomized only 100 patients in its three arms. The confidence intervals were wide and high-flow oxygen is resource intensive.
The French research team, led by the first author Jean-Pierre Frat, must have felt that high-flow oxygen needed stronger evidence. This is commendable because the medical literature is replete with small studies with large effect sizes that are subsequently overturned by larger studies.
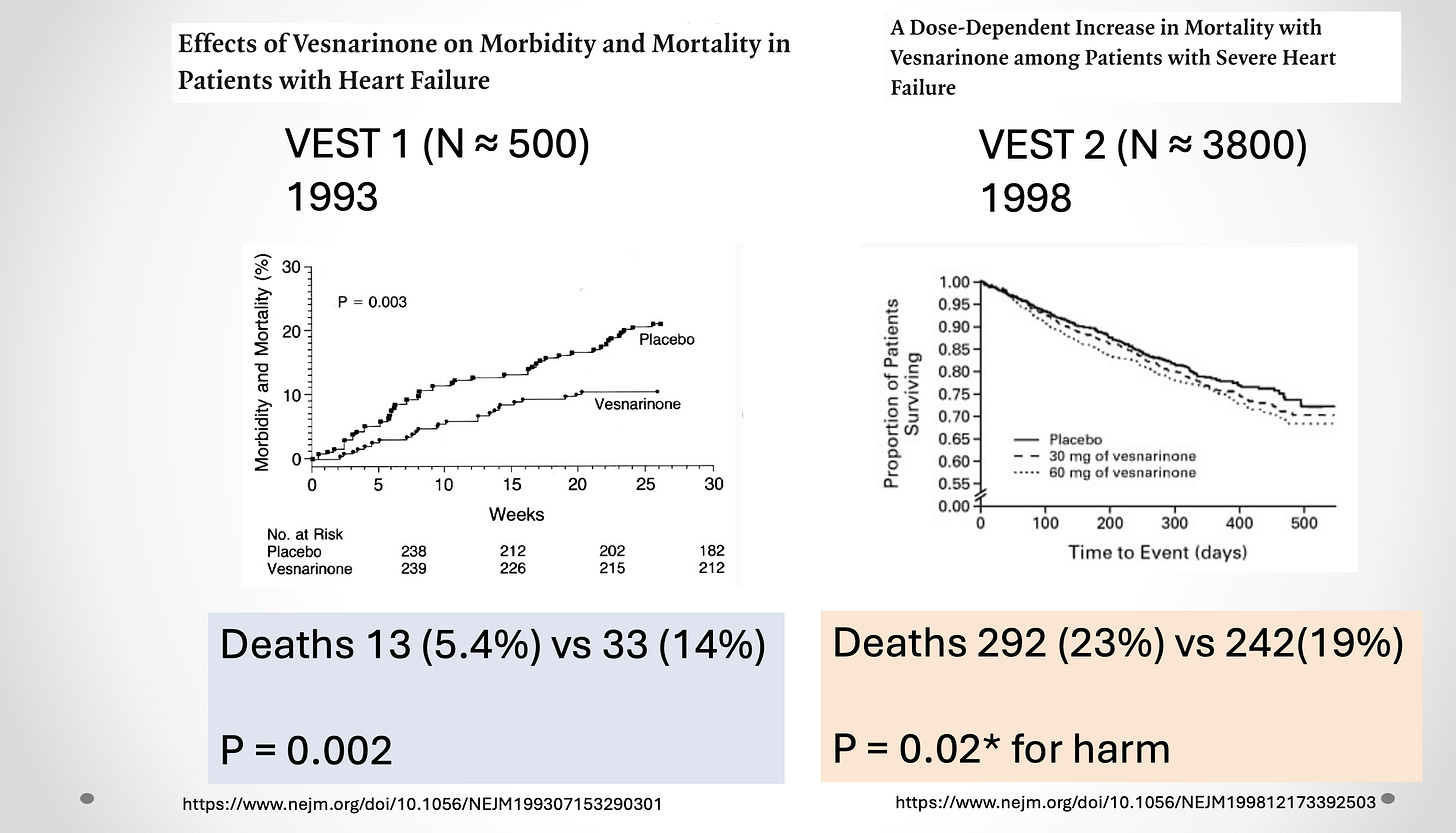
I love to show the positive inotropic drug vesnarinone story
Enter the SOHO study published this month in the NEJM. High-flow vs standard oxygen in 1116 patients with respiratory failure in French ICUs.
The primary endpoint was death at 28 days. No bias there. Alive or dead. Patient selection criteria were strict. You could not have ventilatory issues; pCO2 > 45 was an exclusion as was many other primary lung, cardiac, renal disorders. Most of these patients had viral or bacterial pneumonias and nearly 90% had bilateral infiltrates.
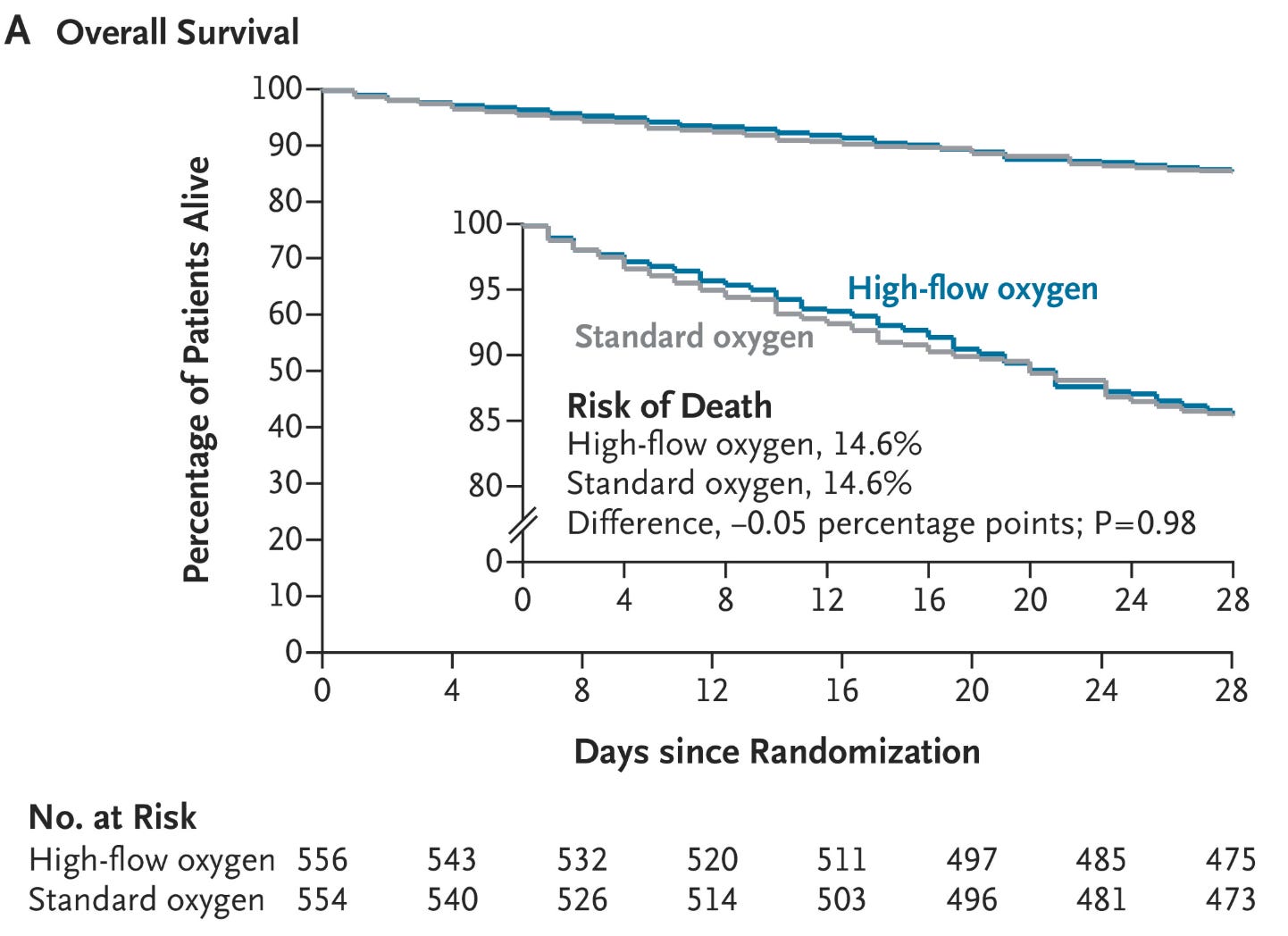
The primary endpoint was as null as null can be: 14.6% (81 of 556 patients) in the high-flow-oxygen group and 14.6% (81 of 554 patients) in the standard-oxygen group (absolute difference, −0.05 percentage points; 95% confidence interval [CI], −4.2 to 4.1; P=0.98).
So, I ask, can this be a short post? Is the chapter closed? Let’s reverse the guidelines, save the resources and use standard oxygen.
Not so fast. As an outside observer to ICU medicine, I found the list of secondary outcomes remarkable.
The incidence of intubation: better in the high-flow group (42% vs 48%* significant)
Median number of ventilator-free days: better in the high-flow group (28 vs 26 days* significant)
Respiratory rate (1-hour after starting): better for high-flow (26 vs 29 breaths/min* significant)
Patient perceived dyspnea: better with high-flow (49.5% vs 34.6%* significant)
Comments:
I am not sure I have seen many studies where the secondary outcomes were more relevant than the mortality outcome.
Although high-flow oxygen did not improve mortality rates, it resulted in less intubation, more time off ventilators and better patient comfort. That’s a lot of benefit.
The caveats include that there were 10 vs 4 cases of pneumothorax in the high-flow arm. This higher rate did not reach significance.
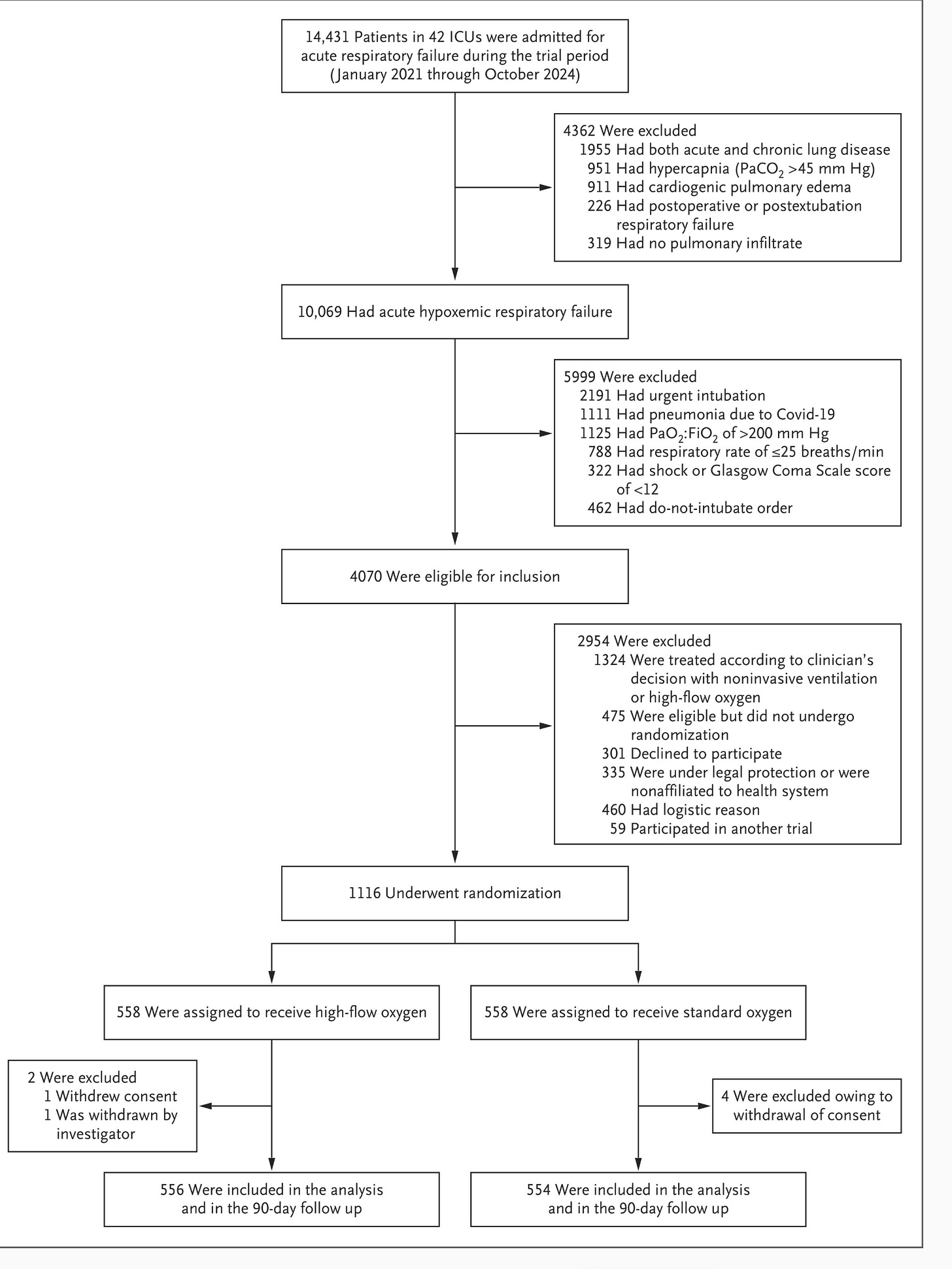
The screened vs enrolled flow diagram is also worth a look.
Kudos to the authors because this is an exemplary flow chart.
More than 14k patients were admitted to 42 ICUs. 10K had respiratory failure with low oxygen. Nearly 6000 of these were excluded for various reasons, such as the need for urgent intubation or shock. This left 4000 patients eligible for the trial. But only 1116 of these were enrolled. More than a thousand were excluded due to clinician’s preference for high-flow oxygen. This is what happens when a therapy gets established. A jargon-like name is erosion of clinical equipoise, which can occur when practices are established on weak data.
The classic example of erosion of clinical equipoise was antiarrhythmic drugs after MI. At the time of the CAST trial, doctors felt it was unethical to randomize in the trial because their patient could get placebo. But placebo had a manyfold better chance of being alive!
Back to the results: Perhaps overall mortality should be demoted to a secondary endpoint in some trials. While it is the most important and only non-biased endpoint, there are often other outcomes on causal pathways of interventions.
For stroke prevention therapies, we look at stroke. For MI prevention studies, we look at composite endpoints including CV death MI and stroke.
Perhaps in respiratory failure studies, we should also look at composite endpoints as well. Let me know what you think?



Thank you John , perceptive as usual
We treat patients for two reasons : to feel better and/or live longer ( note the two do not always go in the same direction).
In very ill patients , living longer is not necessarily the most important outcome . John Spertus and his team illustrated this in an elderly cohort, who asserted ( summarizing) that freedom from disability was MUCH more important than prolongation of life ( my emphasis) .
Our primary purpose as caregivers is to relieve suffering .
Perhaps so. But surrogate endpoints exactly serve the Big Pharma cartel in every example I can think of, in cancer treatment, hypertension, CVD, on and on.
I am not treating patients. But I assume and believe that treating patients involves making them more comfortable.
But in the world of health research, all cause mortality is by far the most important factor and the one that Big Pharma inevitably prefers to squirm out of.
For instance, cancer treatments are sold with the promise that they postpone recurrence and lower death from cancer. That they don’t lengthen lifespan, or that they increase morbidity from side effects, is under reported.
CVD: statins are sold with the idea that they lower death from heart attacks. Their numerous bad side effects and the fact that they don’t extend lifespan is under reported.
Etc. This is just business as usual for Big Pharma.
So you bring up a narrow example that involves patient comfort and show a small but perhaps significant difference in all-cause mortality but I think this example is perhaps not particularly important. If it makes patients more comfortable to have oxygen delivered a different way, then wouldn’t it just be good medicine to deliver oxygen that way?
They used to deliver oxygen with a small amount of CO2, for many good reasons. So-called carbogen fell out of favor but perhaps it should be brought back. Patients are often over oxygenated, lowering their CO2 to dangerous levels.