At ACC26, the HI-PEITHO Trial is in a Photo-Finish for Greatest Spin
Pulmonary embolism is a life-threatening condition that often requires more than anticoagulation. Invasive therapies hold promise but must be held to rigorous standards
I sat in front of the massive audience for the first late-breaking trials session at last weekend’s ACC meeting. I was there to hear the results of the CHAMPION AF trial of Watchman vs anticoagulants. (See last Monday’s post).
CHAMPION AF was the second trial; Hi PEITHO went first. I was only partly paying attention to the trial of an invasive ultrasound-facilitated catheter-directed fibrinolysis treatment of intermediate-risk patients with pulmonary embolism. What caught my ear was the jubilant presenter and discussants. This comment stuck in my brain:
A new era in PE care
I thought to myself: John, you’d better look under the hood of this trial.
Some brief background: a pulmonary embolism occurs when a clot gets lodged in the large pulmonary arteries. If the clot is large enough, oxygen levels drop and the right heart can fail. The mainstay of therapy is anticoagulation, which facilitates the body’s clearance of the clot. When clots are large, and patients sick enough, invasive measures have been proposed for more direct dealings with the clot.
The invasive measure tested in HI-PEITHO is Boston Scientific’s EkoSonic catheter which delivers ultrasound and fibrinolytic therapy right into the clot-ridden pulmonary arteries. The ultrasound is the novel part: it’s supposed to create tiny fluid currents within the clot, thereby disrupting the clot and facilitating the clot-busting fibrinolytic. (I know; it’s a nice story; Medicine overflows with these.)
Stories are nice but they require testing in randomized trials.
The Trial
HI-PEITHO was a multinational open label trial of ultrasound facilitated catheter directed fibrinolysis for intermediate-risk PE patients. Inclusion required an RV/LV ratio >1 and an elevated troponin and two indicators of cardiorespiratory (CR) distress (SBP <110, HR ≥ 100 or RR > 20.
One group gets US-facilitated alteplase plus anticoagulation (AC) and the other gets AC alone. The primary outcome is a composite of PE-related death, cardiorespiratory (CR) decompensation or symptomatic PE recurrence.
We need to talk about the primary endpoint components. PE-related death and PE recurrence are straightforward; CR decompensation was more complex. It included five items: cardiac arrest, signs of shock, placement on ECMO, intubation or initiation of noninvasive mechanical ventilation, or a National Early Warning Score (NEWS) > 9 by two consecutive measures 15 minutes apart.
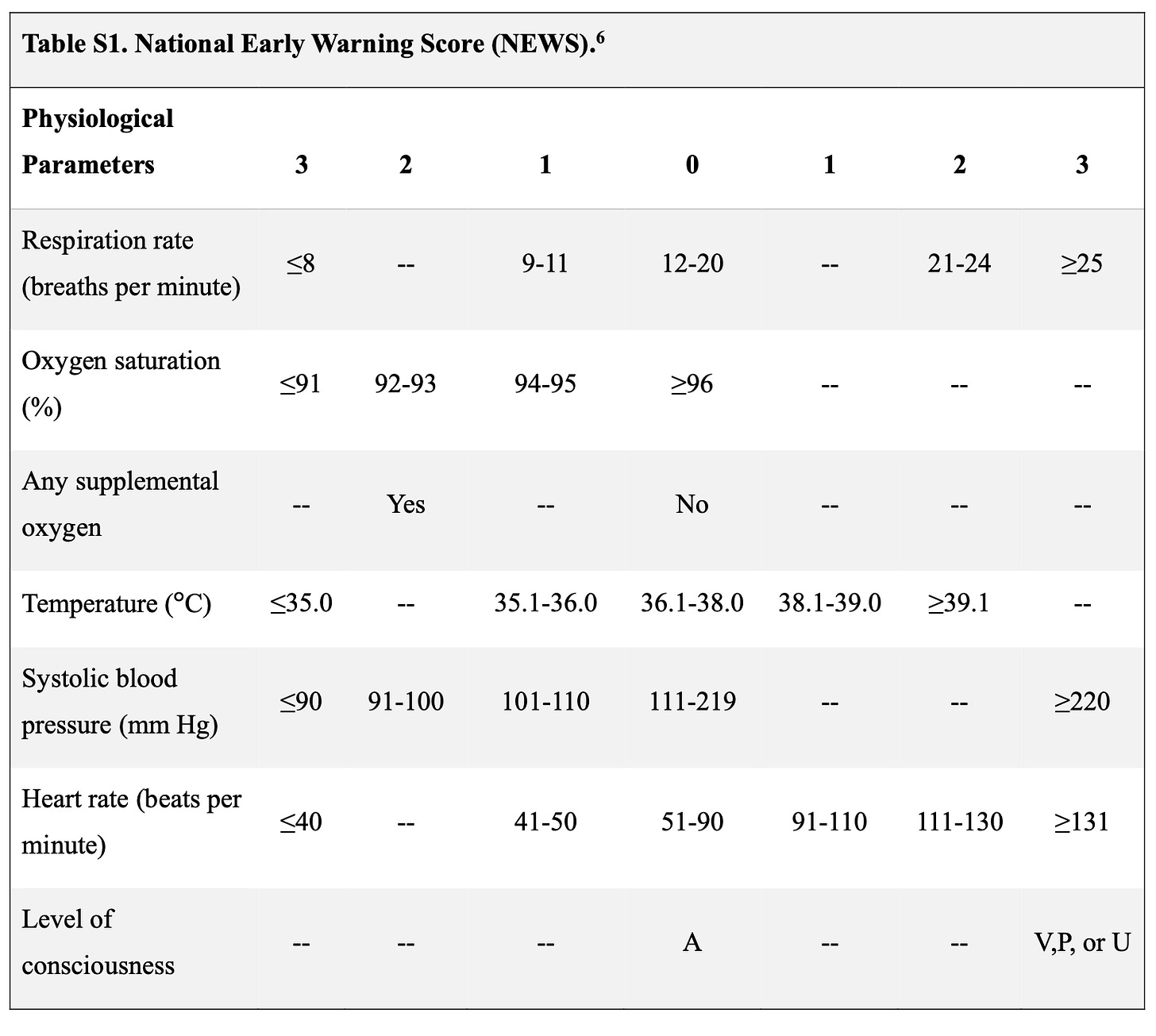
Four of the five components of the CR decompensation endpoint are obvious. But I had never heard of the NEWS score. It’s an ordinal score with each item ranging from 0-3 for bedside physiologic signs including respiratory rate, oxygen saturation, supplemental oxygen use, temperature, blood pressure, heart rate, and level of consciousness. Here is a picture:
The key aspect of this score is that a clinician (possibly a research nurse) makes these judgements, such as respiratory rate of 19 vs 21. I looked up the NEWS score and learned that it was designed as a surveillance and escalation trigger rather than an endpoint for trials. One more fact: HI-PEITHO was open-label so clinicians knew the patients treatment assignment.
The authors screened 4300 patients and enrolled only 544. The maker of the EkoSonic endovascular system, Boston Scientific, sponsored the trial and had representation on the executive committee. Co-sponsors include the University of Mainz and the PERT consortium—which is a nonprofit receiving money from industry. The statistical analyses were performed by a contract research organization, with biostatistics planned by a Boston Scientific employee.
Results
The two arms had about 270 patients.
A primary-outcome event occurred in 11 patients (4.0%) in the intervention group and 28 (10.3%) in the control group (relative risk, 0.39; 95% CI, 0.20 to 0.77; P=0.005).
The drivers of the primary.
PE related death higher in the active arm (3 vs 1)
Recurrent PE were each 1
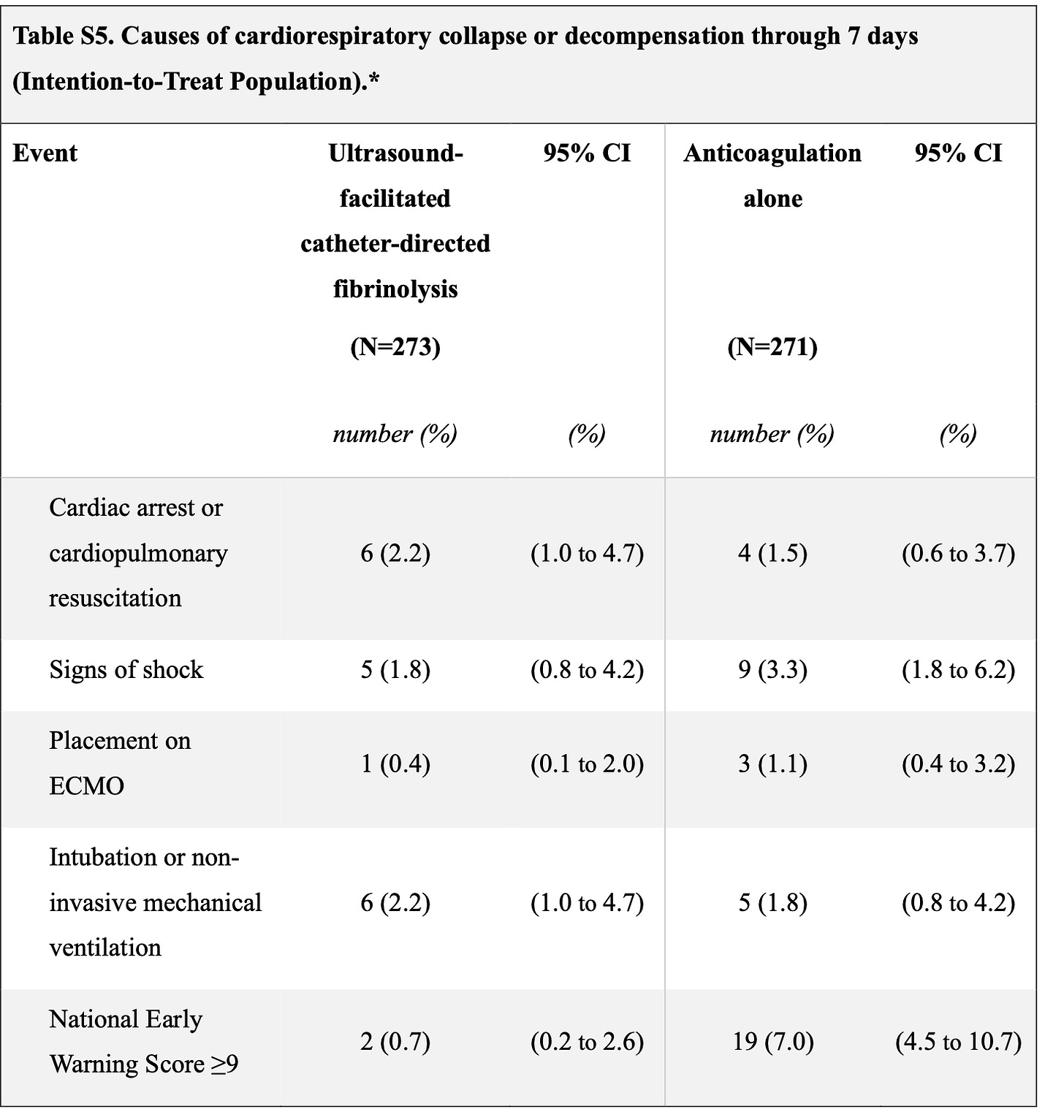
All the difference was lower rates of CR decompensation in the active arm 10 vs 28
So let’s look into the supplement and see about this 10 vs 28 difference.
Cardiac arrest and CPR 6 vs 4
Signs of shock better 5 vs 8
ECMO 1 vs 3
Intubation 5 vs 6
NEWS score > 9 2 vs 19
The primary endpoint was completely driven by bedside score of items judged by a human clinician, like level of consciousness, taken twice 15 minutes apart. Hard outcomes, such as PE-related death was higher, and there was no difference in PE recurrence.
14 of the 19 patients in the control arm who had [NEWS > 9] had only that as the endpoint.
On safety, major bleeding at 72 hours was higher in the Ekosonic arm [3.7% vs 1.5% (RR 2.5, 95% CI 0.8–7.9, p=0.17)]. The p-value is non-significant but there were 2.5x more bleeds in the intervention arm. It used thrombolytics, so it should not be a surprise nor cast off as noise.
At 30 days there were 6 deaths in the invasive arm vs 3 in the control arm. There was also no difference in the 6-minute walk test was (405 vs 393 meters).
The authors declared victory and the discussants called it a new era in PE care. The editorialists wrote that this trial “represents a major advance in the foundation of evidence for thrombolytic approaches.”
Comments
I am not sure why there was so much celebration.
The “positive” result is driven by a soft physiologic score (NEWS >9) in an open label trial. All hard endpoints were clearly not different and trended against the intervention.
Bleeding was 2.5x higher in the invasive arm.
The trial enrolled a highly selected population where 87% screened were not enrolled.
There was no overall mortality benefit and if anything, numerically went the wrong way for the EkoSonic catheter.
Functional outcomes were also nearly identical.
To be fair. PE trials are very hard to do, because presentation is often variable, and, by nature the selection has to be rigorous or you stand a chance to have too much noise—that is, patients who are not sick enough to benefit or too sick to benefit.
That said, however, the lack of signal of improvement in hard outcomes stands out. The driver of the positive endpoint was a prediction score that requires clinician judgement. Clinicians, in this case who are both proponents of invasive measures and aware of treatment assignment.
I would not call this a win for ultrasound directed fibrinolysis. Not at all. If I were a regulator I would reject this therapy.
I mention this trial here on Sensible Medicine because it is another great example of looking deeper than topline results and studying the supplement.
There is also a written review and podcast of this trial over at Cardiology Trials Substack


Is there any way we can ask our colleagues to stop this trend of standing ovations for trials? Most of the positive trials these days are incremental (and many are worse). Sure the teams should be proud of what they are doing, and I commend them for working on the trials, but it is turning out profession into a circus/clown show. Let's get back to serious science please.
Medicine really needs you and your careful and honest evaluation when industry and over enthusiastic investigators are so keen to ignore the real facts and just sell a new procedure or in this case an expensive device. As with the Left atrial plugs this technique will be adopted by enthusiasts despite the lack of evidence - again the real evidence is that it probably does more harm than good. One hopes that NICE in the U.K. will evaluate with more care and avoid giving it a licence.