Last week I saw the end result of a biased trial, lax regulation, absent peer review and groupthink
I start this review of the OPTION trial with an apology to Sensible Medicine readers.
I am sorry to have not posted on this trial of left atrial appendage closure (LAAC) after ablation for patients with atrial fibrillation (AF) when it came out in November of 2024. I did write two pieces on it over at Medscape Cardiology. The first of these I wrote before the trial results were presented wherein I alerted readers to the biased design and likelihood of positive results. I was 100% accurate.
OPTION re-entered my brain because it was mentioned so heavily at the Western AF meeting that I attended in Salt Lake City this weekend. The trial results had clearly influenced a number of doctors to do concomitant appendage closure as a routine. I even sat on a panel that was entitled “which patient do you do concomitant LAAC in?” My take was no patient, but I was the only outlier.
Let me tell you about this study and then you can decide.
Some background: LAAC is felt by most of my colleagues (not me) as a strategy to mitigate stroke risk and reduce bleeding. The idea is to place a device into the left atrial appendage to stop clots from forming. Then you can stop oral anticoagulation (AC). Patients love the idea.
I recently reported on the CLOSURE AF trial, a non-industry funded German study that showed LAAC was clearly inferior to oral AC.
A seminal trial called PREVAIL found that the device failed to make non-inferiority to warfarin in its first co-primary endpoint of stroke, systemic embolism and cardiovascular death.
Nonetheless, FDA approved it in 2016, and it has become a popular treatment strategy. You may have seen the direct-to-consumer ads.
The Trial
The OPTION trial investigators and funders tested the concomitant use of LAAC at the time of AF ablation as an alternative to ablation and oral AC. The standard of care is to continue oral AC after ablation and LAAC could possibly be a mechanical solution to stroke prevention.
Exactly 1600 patients were randomized to either oral AC or LAAC after AF ablation. The average age was 69 years; a third were female and the stroke risk moderate at an average CHADSVASC score of 3.5. Follow-up was three years
Pay close attention to the choice of endpoints because it is a huge source of bias. The primary safety endpoint (tested for superiority) was a composite of non-procedural bleeding or clinically-relevant nonmajor bleeding. (Pause here for another background fact: the most common procedural complication from LAAC is bleeding into the sac of the heart called pericardial tamponade. It most often requires either a chest tube to drain the blood or surgery).
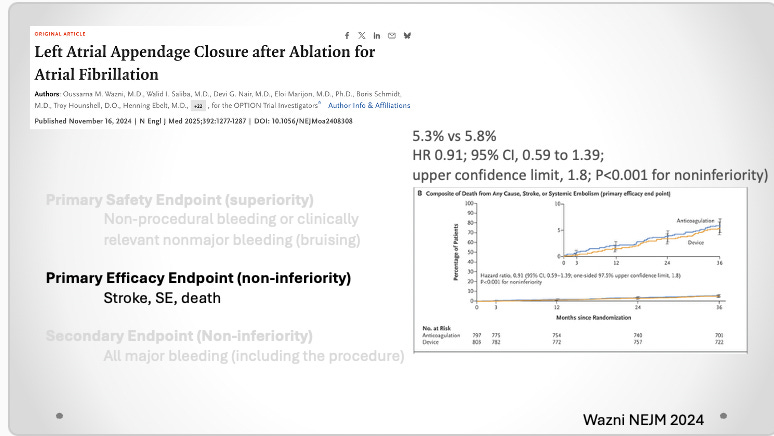
The primary efficacy endpoint (tested for non-inferiority) was a composite of stroke, systemic embolism and death. (Pause here for another background point: it has been established that neither oral AC nor LAAC has any effect on all-cause death. See comments.)
The secondary endpoint (non-inferiority) was major bleeding including the procedure.
I will show the results in slides I presented in my talk at Western AF.
Here is the primary safety endpoint. The 56% reduction of non-procedural bleeding or nonmajor bleeding strongly favored LAAC.
Here is the primary efficacy endpoint. The similar rates of stroke, systemic embolism and death met criteria for non-inferiority.
Here is the secondary endpoint of all major bleeding including the procedure. Note that it makes non-inferiority but does not meet superiority criteria.
The authors made positive conclusions in the NEJM and what I witnessed at Western AF convinced me that it had had an effect. Doctors are doing this procedure at the time of AF ablation.
Yet I see this as one of most biased trials ever done in my field of electrophysiology. Perhaps you see the bias too.
Primary Safety Endpoint Flaws
There are two huge flaws in the choice of the primary safety endpoint. One is that they excluded procedural bleeding. This defies all clinical sense because patients cannot exclude procedural bleeding. While inadvertently poking a hole in the left atrium during the procedure has been reduced, it is still a serious life-threatening complication requiring intervention. The second flaw is the inclusion of nonmajor bleeding as an endpoint in a non-blinded trial. In OPTION, patients knew their treatment assignment; blinding was not feasible. So patients on oral AC are more likely to report nonmajor bleeding. Indeed, things like bruising, lacerations, and oral bleeding were higher in the AC arm. (Event rates were 42 vs 104)
Primary Efficacy Endpoint Flaws
This is slightly more nuanced but just as important. Efficacy of a stroke prevention strategy is the reduction of stroke and systemic embolism (clot going somewhere other than the brain). The problem for testing this is that these are low-frequency events. That means you need a lot of patients to test true non-inferiority. And that adds cost and complexity to a trial.
The workaround is to add death as an endpoint. Since these are older people, some will die in the three years of the trial. But the problem is that neither oral AC or LAAC affects death. Everyone knows this. A surgical trial called LAAOS 3 showed that surgical LAAC and AC reduced stroke over AC alone but had no effect on death. The reason is that stroke and bleeding are minor causes of death compared to, say, cancer.
The problem with adding an endpoint not affected by either treatment is that it adds noise (more events) and makes no difference easier to achieve. Recall that in a non-inferiority comparison, no difference is a “positive” outcome for the new treatment.
Indeed, that is exactly what happened. Death was the largest driver of this endpoint and was no different. Stroke rates were 1.2% and 1.3%. At that low incidence, it would have taken 5x more patients to sort out true non-inferiority.
The OPTION trial, therefore, was noninformative regarding efficacy due to low statistical power.
Secondary Endpoint Flaw
The secondary endpoint of all major bleeding should have been the primary safety endpoint. Not only that, but in non-inferiority trials of new treatments vs established treatments, the correct analysis is to test efficacy with non-inferiority (just as good), and safety with superiority.
In this case, the all major bleeding was not superior to the standard treatment of oral AC. Hence, LAAC offers no safety advantage over oral AC.
Other Worrisome Signals
In this piece on Medscape Cardiology I identify other worries in OPTION, including missing data, and the fact that 1 in 5 patients in the device arm had peri-device leaks, which are associated with higher rates of stroke.
Conclusion
My conclusion is sadness. My electrophysiology colleagues are highly educated intelligent people. EP doctors understand vectors, physics and biology.
How can you have the brain power to be an electrophysiologist and not recognize a clearly biased and uninformative trial? How could FDA regulators have allowed such a trial? How could medical scientists signed on to do it? And where were the editors and peer reviewers at NEJM?
I am not sure whether this is important: a Canadian electrophysiologist told me that LAAC is a quite uncommon procedure in Canada.




Why do you think no one on NEJM peer review board saw these glaring issues with this study design? It is becoming clear to me that unquestionably accepting information from these previously respectable purveyors of medical discovery is foolish, and also lazy. We owe it to our profession and our patients to do the hard work as you have done, and critically call out these issues. Thank you again for clarifying what seems to be an obvious problem. Perhaps embarrassing these folks, as you have done, can have a positive effect, but something tells me I should not hold my breath waiting.
The real groupthink is ignoring the role of rT3 in a fib. It’s been established beyond any doubt that a fib is connected with high rT3. And many older folks with a fib are routinely prescribed T4 which makes the problem worse.
So I would say physicians are the cause of a lot of a fib and these sorts of studies ignore the basic fundamentals in favor of very expensive interventions.
Physicians are taught to “ablate” — a form of cauterization — instead of treating a fib as a thyroid disorder brought on by patients with poor metabolic rate, high cortisol, and low thyroid function.
I’ve spoken to doctors about this, including an endocrinologist recently, and they all just scoff at this and shut me down even though there is ample evidence.
so-called “sub clinical hypothyroidism” and similar terms mean the same thing. Tuning a patient’s T4-only dose using TSH levels is madness.
Very very few studies, let alone doctors, even bother to test for rT3.
Shame on Big Medicine.
https://pubmed.ncbi.nlm.nih.gov/27857052/
Results: “permanent A fib” group showed higher FT4 and rT3 (1.41 vs. 1.27 ng/dl, p=0.0007; 0.61 vs. 0.32 ng/ml, p<0.0001, respectively). With ROC curve analysis the biochemical thyroid related factor of the highest prognostic value for PAF occurrence (with the highest sensitivity and specificity: 77% and 72%, respectively) was rT3 with the cut-off of above 0.3 ng/ml. Also, a positive correlation between rT3 levels and left ventricular posterior wall diameter was observed (Spearman's correlation coefficient 0.33, p=0.0093).